
Most metals are good conductors of heat and electricity, are malleable and ductile, and are moderate to high melting points. The 14 elements following lanthanum (z=57) are called lanthanides, and the 14 following actinium (z=89) are called actinides.Įlements in the periodic table can be placed into two broad categories, metals and nonmetals. The two rows of elements starting at z=58, are sometimes called inner transition metals and have that have been extracted and placed at the bottom of the table, because they would make the table too wide if kept continuous. The elements shaded in light pink in the table above are known as transition metals. These groups contain the most naturally abundant elements, and are the most important for life. 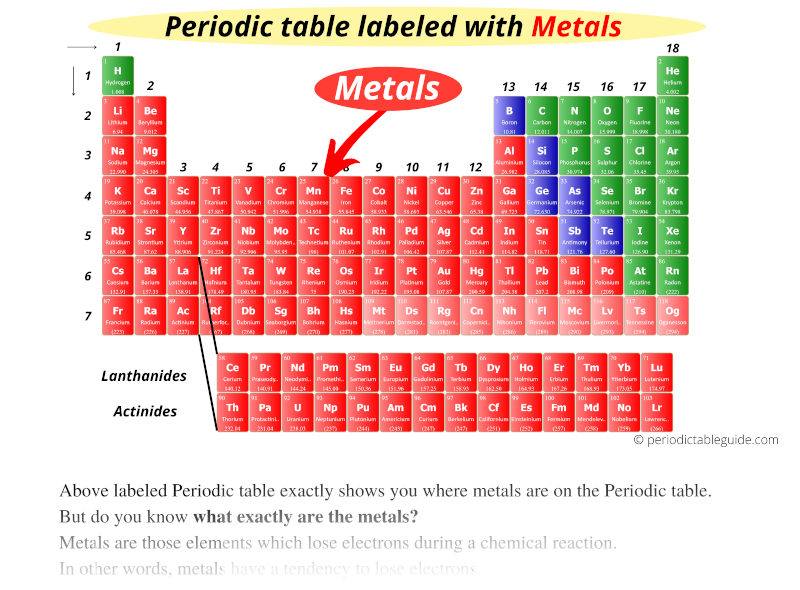 
The main group elements are groups 1,2 and 13 through 18. The groups are numbered at the top of each column and the periods on the left next to each row. From left to right, the atomic number (z) of the elements increases from one period to the next (horizontal). On the periodic table, elements that have similar properties are in the same groups (vertical). Although most modern periodic tables are arranged in eighteen groups (columns) of elements, Mendeleev's original periodic table had the elements organized into eight groups and twelve periods (rows). Mendeleev believed that when the elements are arranged in order of increasing atomic mass, certain sets of properties recur periodically. Dmitri Mendeleev, a Russian scientist, was the first to create a widely accepted arrangement of the elements in 1869. The Periodic Table of Elements categorizes like elements together.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
